Our Technology
A comprehensive solution bridging repair, monitoring, and rehabilitation
AI agents are transforming healthcare. Soon every physician will have the ability to optimize care with data. But only if the data exists.
Surgeries succeed, but the real bottleneck is post-discharge. Once patients go home, the data stream stops. The availability, content, and quality of post-discharge data determines whether "data-driven care" is a slogan or reality.
AI drives software development costs toward zero — anyone can build a rehab app. But non-replicable clinical data is the real moat. Value lives in the data flow, not the code. This is the essence of thick software.
741 hospitals
CMS TEAM mandatory bundled payments, Jan 2026. 30-day episodes + PROM ≥50%, zero infrastructure funding. CMS.gov →
51% same-day discharge
TKA mean LOS dropped from 3.5 to 0.89 days. Patients go home faster — but the monitoring gap widens.
AAOS 2026
RTM validated in geriatric populations — no complication increase, lower costs. Healio →
WingHeal™
Bio-inductive Reinforcement System
Rotator cuff repairs face a re-tear rate of up to 40%. Current solutions rely heavily on mechanical fixation but often fail at the tendon-bone interface.
Furthermore, once the patient leaves the OR, surgeons are "flying blind"—there is no existing solution for wirelessly detecting repair failure before it's too late.
A hybrid solution combining a PEEK augment with a bio-inductive scaffold, designed to enhance the tendon-bone interface and promote biological healing.
+71.8%
Increase in Biomechanical Stiffness — Compared to standard TOE repair in comparative goat models
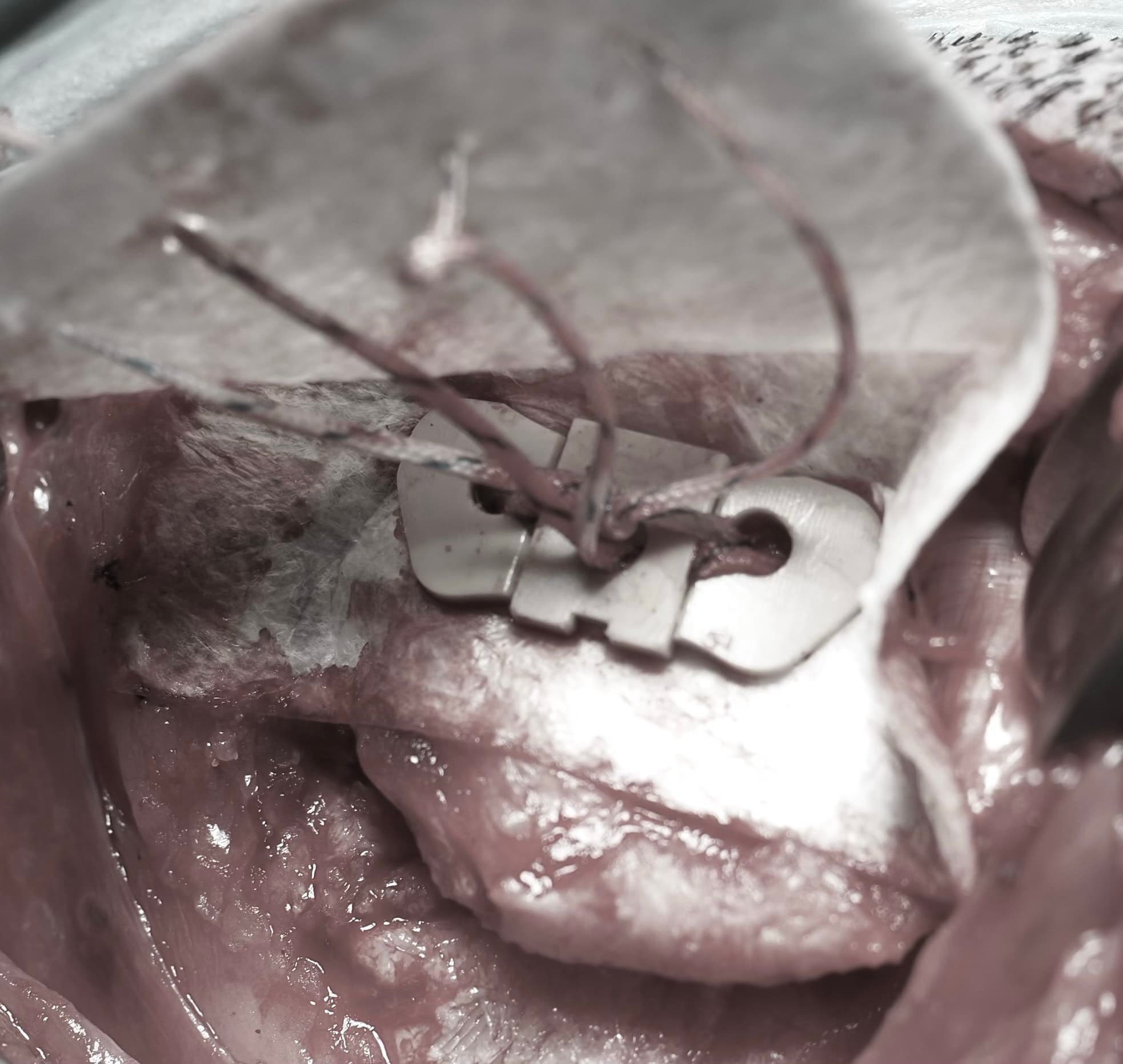
Animal Model Study
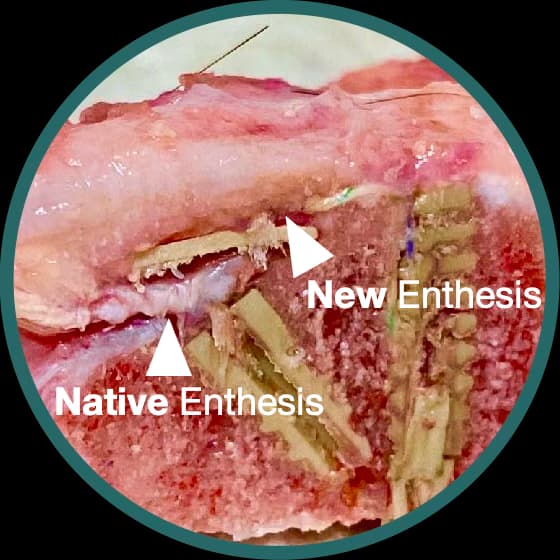
Enhanced Enthesis Growth
- 01PEEK Augment — Mechanical reinforcement at the repair site
- 02Bio-inductive Scaffold — Promotes tissue integration
- 03Cost-effective — Streamlined alternative to complex biological grafts
$1.13B global rotator cuff repair device market (2023).650K+ annual US repairs.
Lin CW, Chiang ER, Chen SH, Chen P, Liu HJ, Chiu JCH. Global Compressive Loading from an Ultra-Thin PEEK Button Augment Enhances Fibrocartilage Regeneration of Rotator Cuff Enthesis. Bioengineering. 2023;10(5).
The regulatory and policy information on this page is based on publicly available sources as of Q1 2026 and is provided for informational context only. It does not constitute legal, regulatory, or medical advice. Policy details and market data may change.
Discovery-R™
Implantable Smart Sensing
Closing the loop on recovery. Discovery-R is a wireless sensor designed to be implanted alongside the repair, providing real-time data on tissue integrity.
It transforms post-op management from "wait and see" to "monitor and act"—enabling the early detection of failure before it's clinically apparent.

Implantable Sensor Module
180 days·2,000 compression cycles
Battery-free passive sensor maintains signal stability under sustained cyclic loading in vivo.
r = 0.87–0.92
Implant gait data correlation with clinical measurement — high predictive value by 6 weeks post-op.
Internal data, investigational


The regulatory and policy information on this page is based on publicly available sources as of Q3 2025 and is provided for informational context only. It does not constitute legal, regulatory, or medical advice. Policy details and market data may change.
iRehab™
LiveContinuity of Care
The digital bridge connecting surgery to full recovery. iRehab gives patients a guided daily routine and gives surgeons real-time visibility into how every patient is healing — no extra clinic visits required.
Already live in clinical settings. In the future, iRehab will integrate real-time data from Discovery-R implantable sensors to enable truly personalized, data-driven rehabilitation.

Clinical Evidence
20 RCTs · 3,706 patients
Telerehab is non-inferior to in-person rehab — no significant difference in pain or function.JMIR 2025 →
53.85% vs 9.5%
Electronic PROM completion is 5.7x paper. Multi-channel (SMS + App) reaches 85%+.
65–85% adherence
Digital rehab adherence significantly exceeds traditional home exercise programs (40–60%). Structured guidance beats a printed sheet.
93.7–99% satisfaction
Patient satisfaction after actual use. Objections come from unfamiliarity, not inability.
- Recovery Engine — 34 evidence-based exercises across 5 clinical phases — from prehab to return-to-function. Adaptive morning/evening sessions keep patients on track without overwhelming them.
- PROM Collection — QR-code-first functional outcome surveys (PROMIS, KOOS JR, HOOS JR, QuickDASH). Achieving over 50% matched baseline-to-discharge completion rates.
- Clinical Alerts — Surgeon dashboard flags low adherence, rising pain trends, and inactive patients in real time — so intervention happens before complications.
- Wound Photo Tracking — Daily photo calendar with streak gamification. Patients document healing progress; surgeons see the timeline without scheduling extra visits.
- Phase Governance — Clinicians control phase advancement — the system never auto-promotes. Skip-with-reason tracking reveals why patients struggle, not just that they do.
- Care Coordination — Magic-link access (no passwords), automated milestone celebrations at day 3/7/14/21/28, and one-tap LINE sharing keep patients and care teams connected.
The regulatory and policy information on this page is based on publicly available sources as of Q1 2026 and is provided for informational context only. It does not constitute legal, regulatory, or medical advice. Policy details and market data may change.
Interested in Our Technology?
We welcome discussions about clinical partnerships, research collaboration, or investment opportunities.
Contact UsCAUTION
WingHeal and Discovery-R are investigational devices. Limited by law to investigational use. Not available for sale.